HOME | AKTUELLES | TEAM | FORSCHUNG | PUBLIKATIONEN | LEHRE | STELLENANGEBOTE
Research
Molecular mechanisms of Adhesion GPCR functions and their modulation
Adhesion G protein-coupled receptors (aGPCR) are functionally and structurally highly diverse receptors displaying unique structural features that set them apart from other GPCRs. They are often described as hybrid molecules fulfilling dual roles in adhesion and signalling. Their most striking features are their extraordinarily long N termini encompassing various domains associated with cell-cell or cell-matrix adhesion. Via these extracellular entities, which are autocatalytically cleaved in some Adhesion GPCRs, these receptors mediate different functions: classical G protein signals, adhesion, but also functions solely dependent on the N terminus, which are highly unusual for GPCRs. We found that Adhesion GPCR even convey signals through their N termini (trans function) in addition to the classical G protein signals (cis function) (Fig. 1).
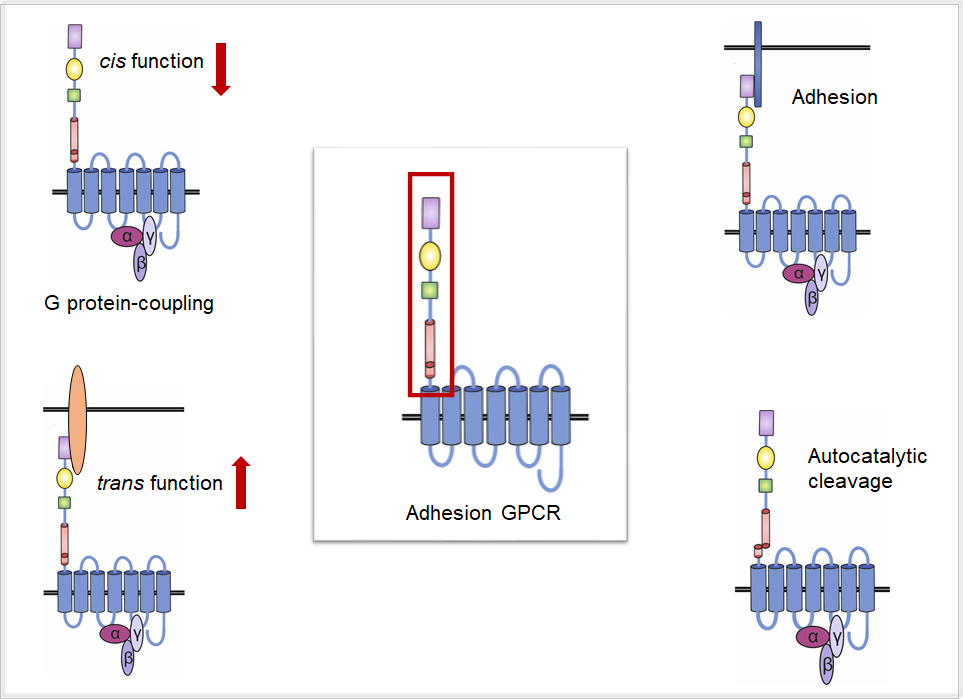
One main focus of our work is to understand how Adhesion GPCRs mediate these numerous molecular functions and how the extraordinarily long and complex N termini integrate them in a tissue- and cell type-specific manner. For this purpose, we employ cellular, genetical, biochemical and pharmacological analyses to investigate expression and signalling properties of the receptors. Further, we aim to identify and characterise interaction partners of the N termini as well as molecules activating the receptors in different contexts and map their binding properties to gain insights on the cis versus the trans function. Interestingly, the repertoire of many Adhesion GPCRs is enlarged by the occurrence of splice variants, yielding a plethora of yet to be investigated variants, which might be one way of realising different receptor functions. To elucidate these variants, their properties and physiological impact, we use structure-function analyses in various cellular contexts as well as in vivo in the roundworm C. elegans.
Adhesion GPCRs in cellular processes
The molecular functions of Adhesion GPCRs have an immediate impact on cellular shape and characteristics, which form the basis for cellular responses to Adhesion GPCR activation. This is especially the case as several Adhesion GPCR have been shown to bind to extracellular matrix proteins and also modulate cytoskeletal rearrangements. We aim to characterise the impact Adhesion GPCRs have on the cytoskeletal components as well as the extracellular matrix and the underlying mechanisms in vitro in biochemical and cellular analyses and in vivo in the nematode C. elegans (Fig. 2).

The role of Adhesion GPCRs in cell polarity, proliferation and development

Many functional features of Adhesion GPCRs such as the ability to mediate cellular adhesion and to transduce signals at the same time make them prime candidates for being involved in cell division and polarity processes. Multicellular organisms are highly dependent on their cells to be spatially organised to form different tissues and organs and to develop/maintain specialised functions (Fig. 3A). For this organisation, cells require a sense of orientation and a form of polarity. Various types of polarity are involved in realising these processes and it is becoming increasingly clear that Adhesion GPCRs are indeed vital players in these.
We investigate the detailed roles of Adhesion GPCRs in cell division and polarity as well as the mechanisms underlying their functions in vivo. One group of Adhesion GPCR involved in
the control of cell division and essential for cell polarity are Latrophilins. The Latrophilin homologue LAT-1 in C. elegans is a regulator for oriented cell division in the early embryo (Fig. 3B) as well as for correct cell division (Fig. 3C). Further, we study mammalian homologs which seem to be involved in the regulation of cell division during development.
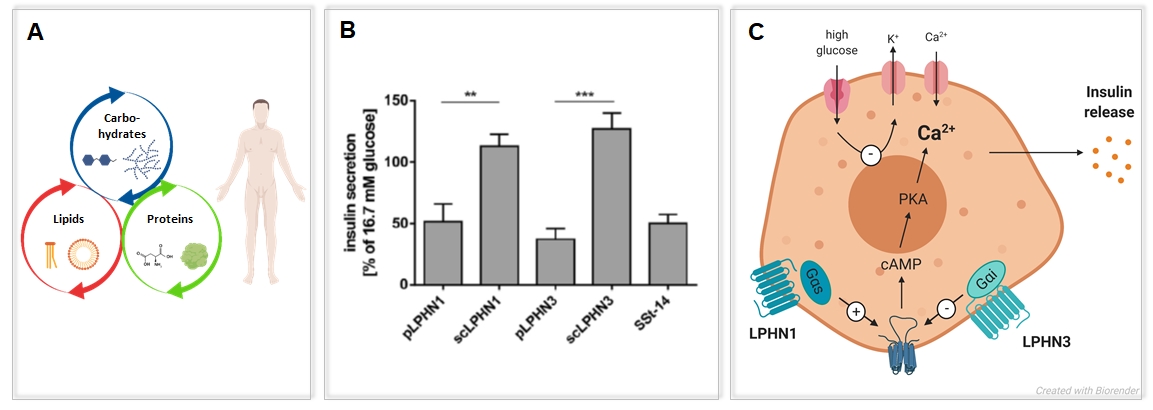
Functions of Adhesion GPCRs in metabolic processes
Despite the increasing evidence that Adhesion GPCR are essential in health and disease their pharmacological potential remains elusive. Especially their possible roles in metabolic processes makes them interesting therapeutic targets (Fig. 4A). Several Adhesion GPCRs have implications in the control of metabolic processes.
We aim to uncover their physiological functions in regulating fat and glucose metabolism as well as kidney function using different model organisms. Thereby, one main focus is on the Adhesion GPCRs Latrophilins, GPR110, GPR111 and GPR115, which have for instance been shown to regulate insulin release (Fig. 4B, C) or liver metabolism.